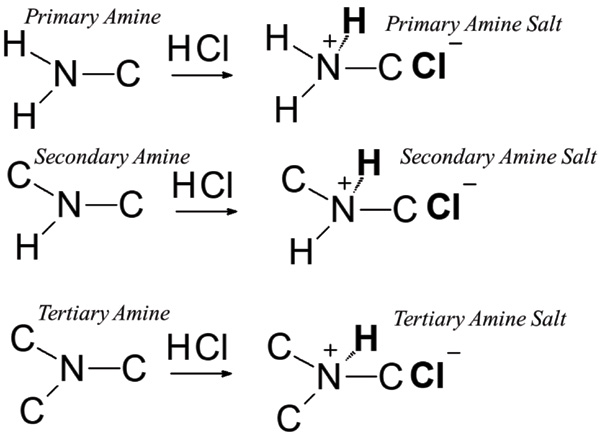
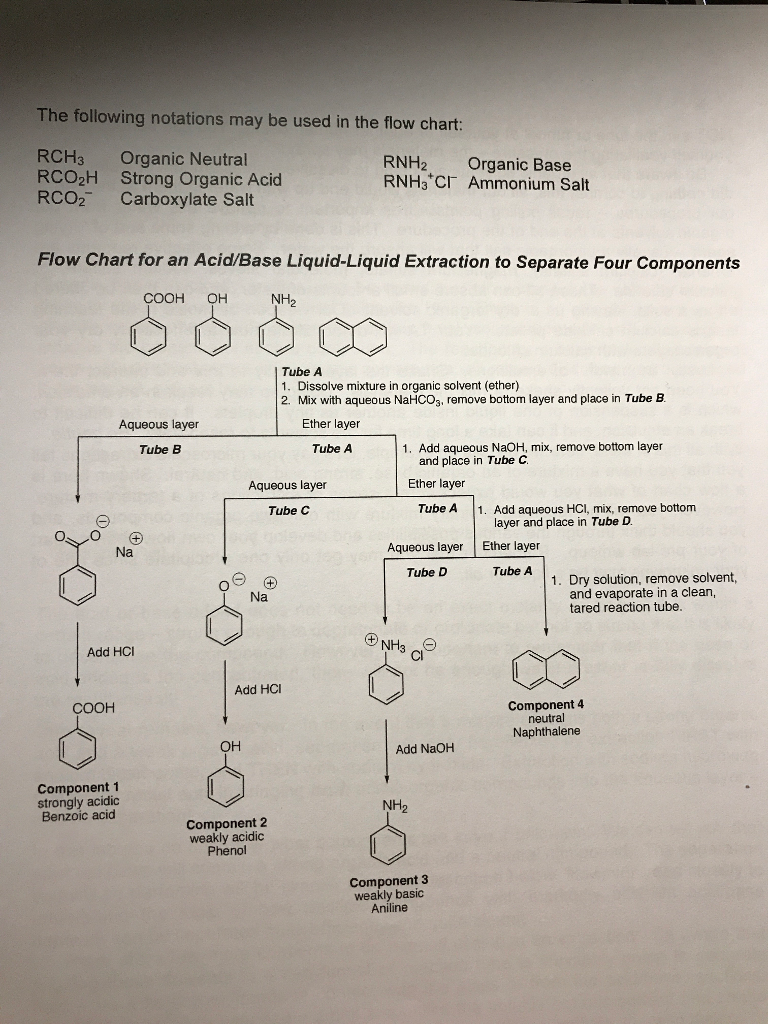
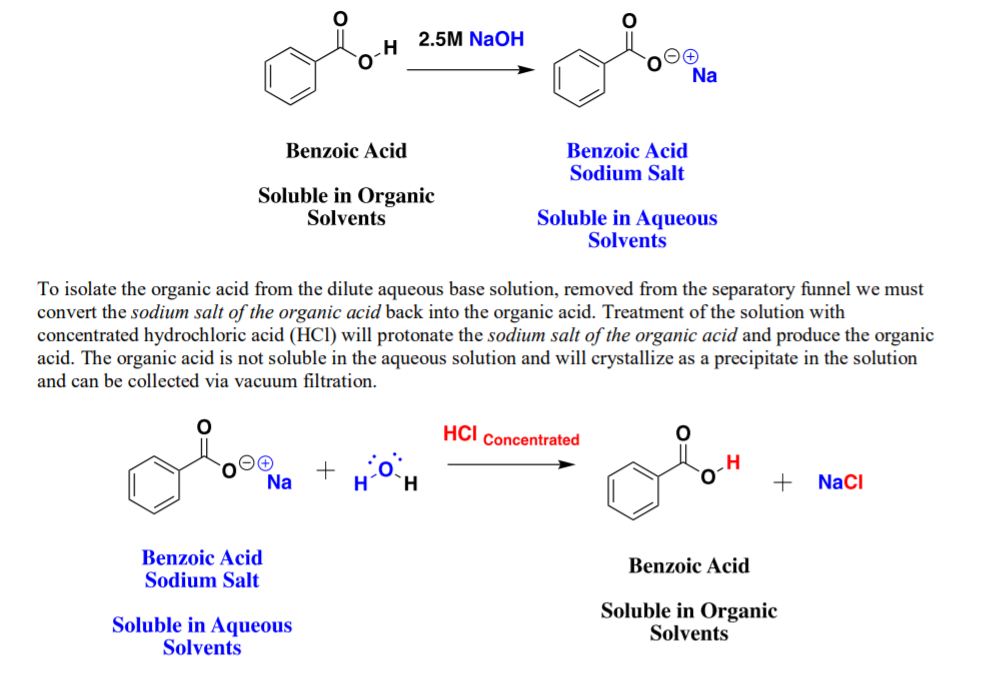
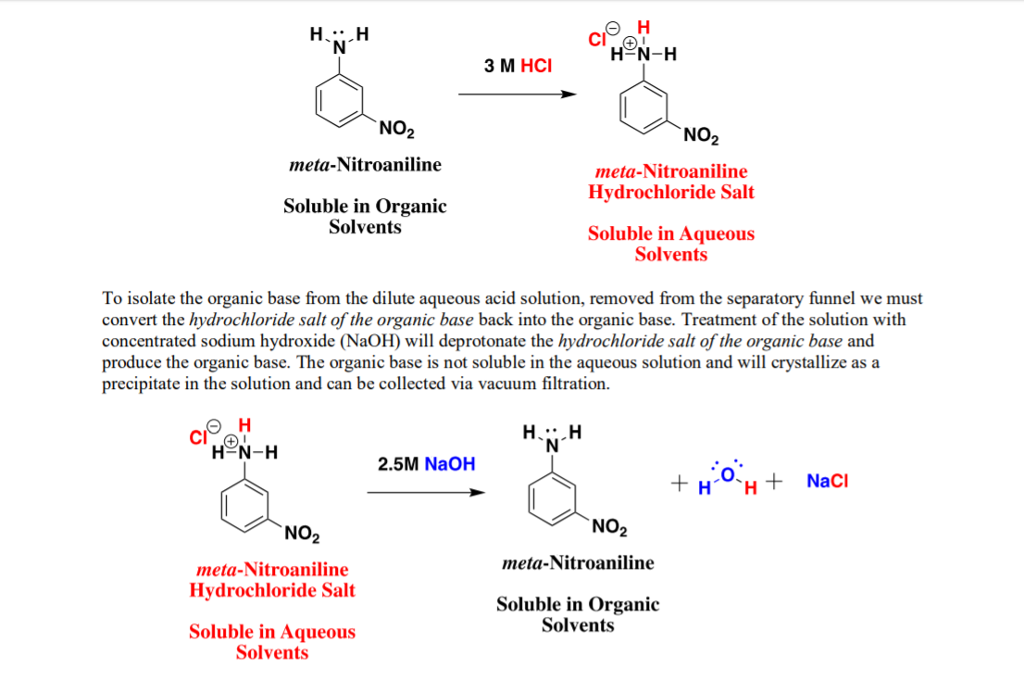
Sodium salt of an organic acid 'X' produces effervescence with conc.H2SO4 . 'X' reacts with the acidified aqueous CaCl2 solution to give a white precipitate which decolourizes acidic solution of KMnO4 'X'

A Bronsted Organic Acid Forming Solid State Salts with Chemically Stable Cationic Species | TCI EUROPE N.V.
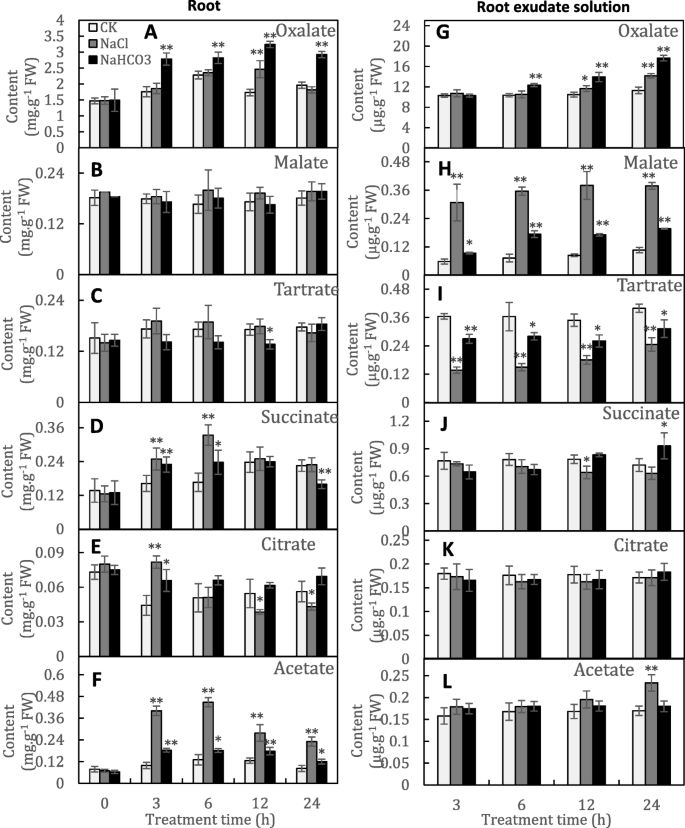
Transcriptomic and phosphoproteomic profiling and metabolite analyses reveal the mechanism of NaHCO3-induced organic acid secretion in grapevine roots | BMC Plant Biology | Full Text
Sodium salt of an organic acid 'X' produces effervescence with conc. H2SO4. 'X' - Sarthaks eConnect | Largest Online Education Community
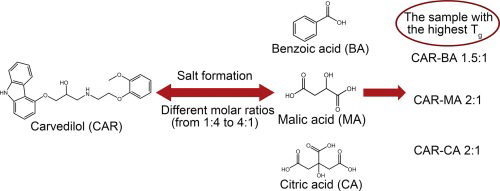
Organic acids as co-formers for co-amorphous systems – Influence of variation in molar ratio on the physicochemical properties of the co-amorphous systems - pharma excipients