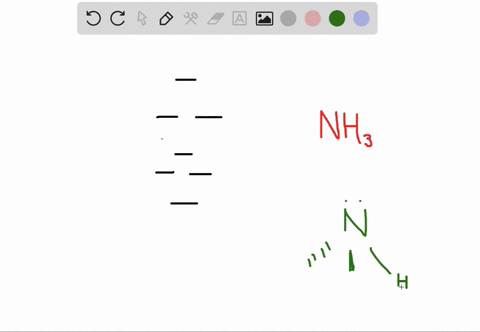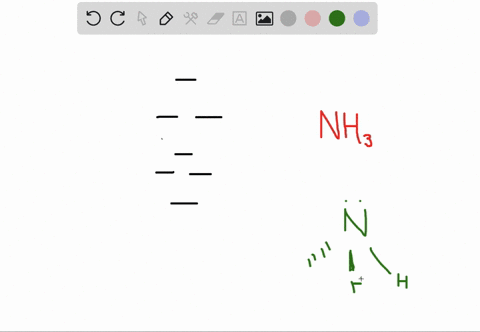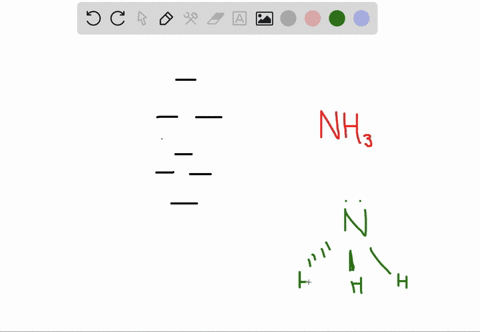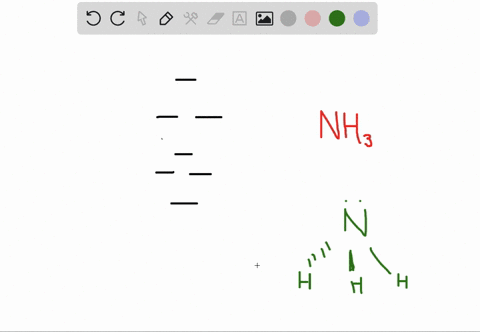
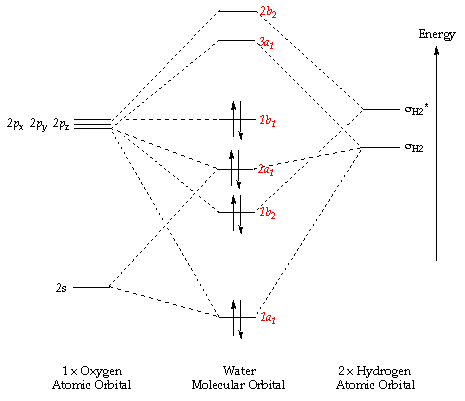
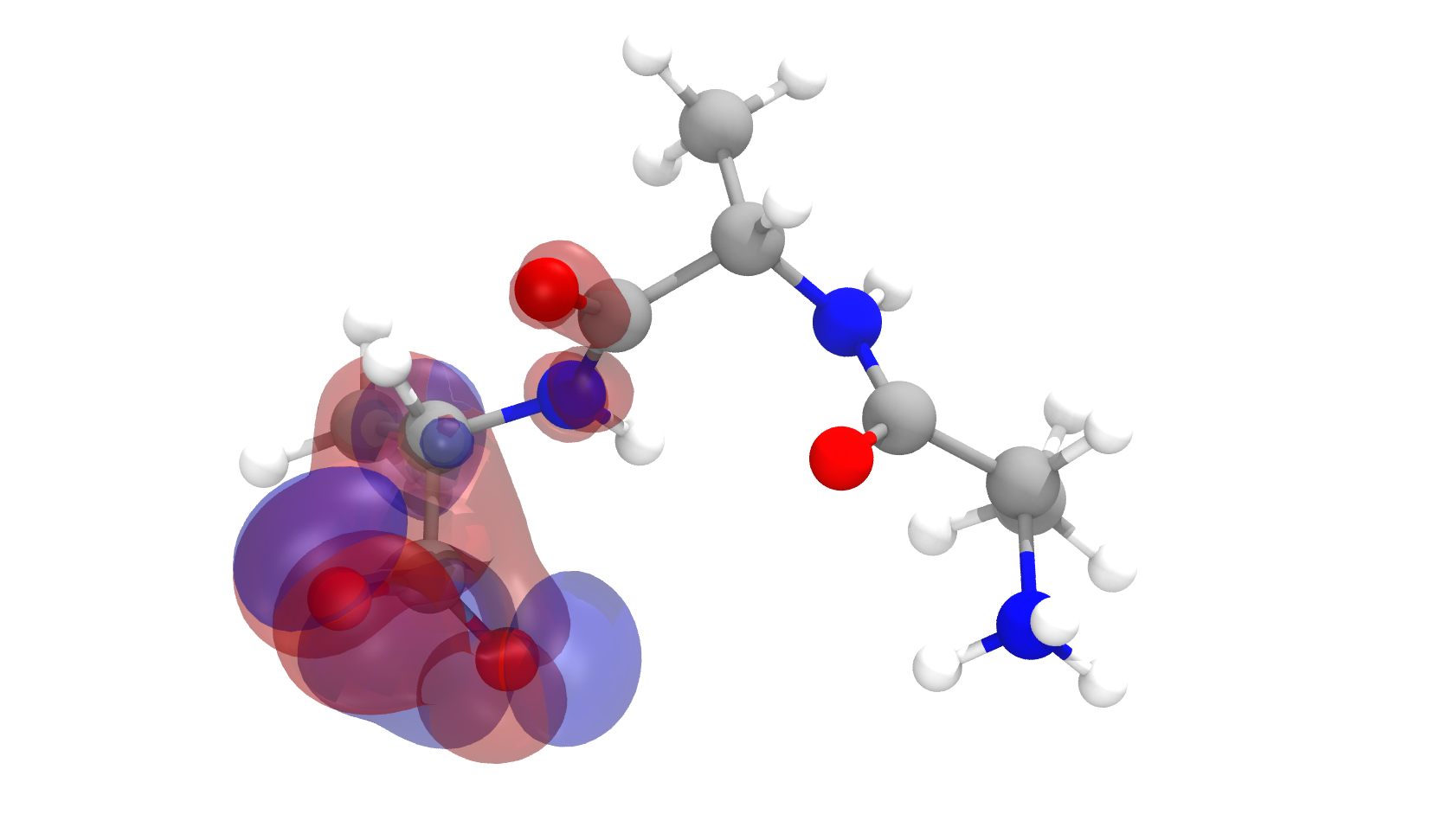
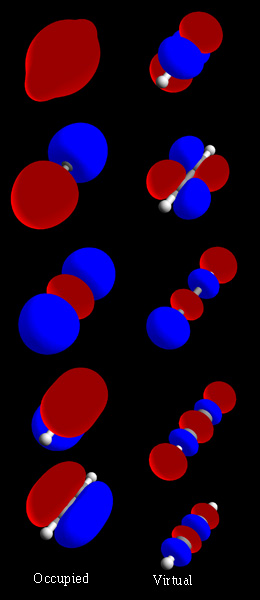
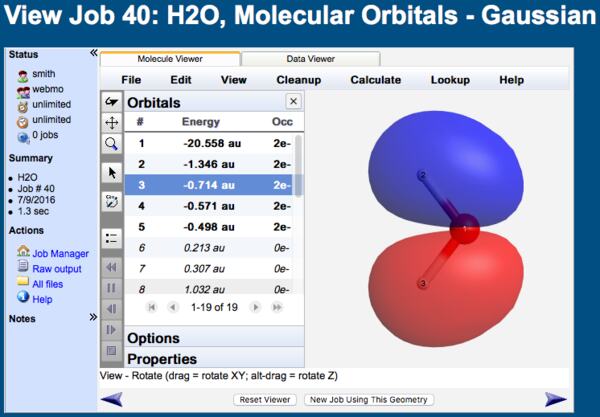
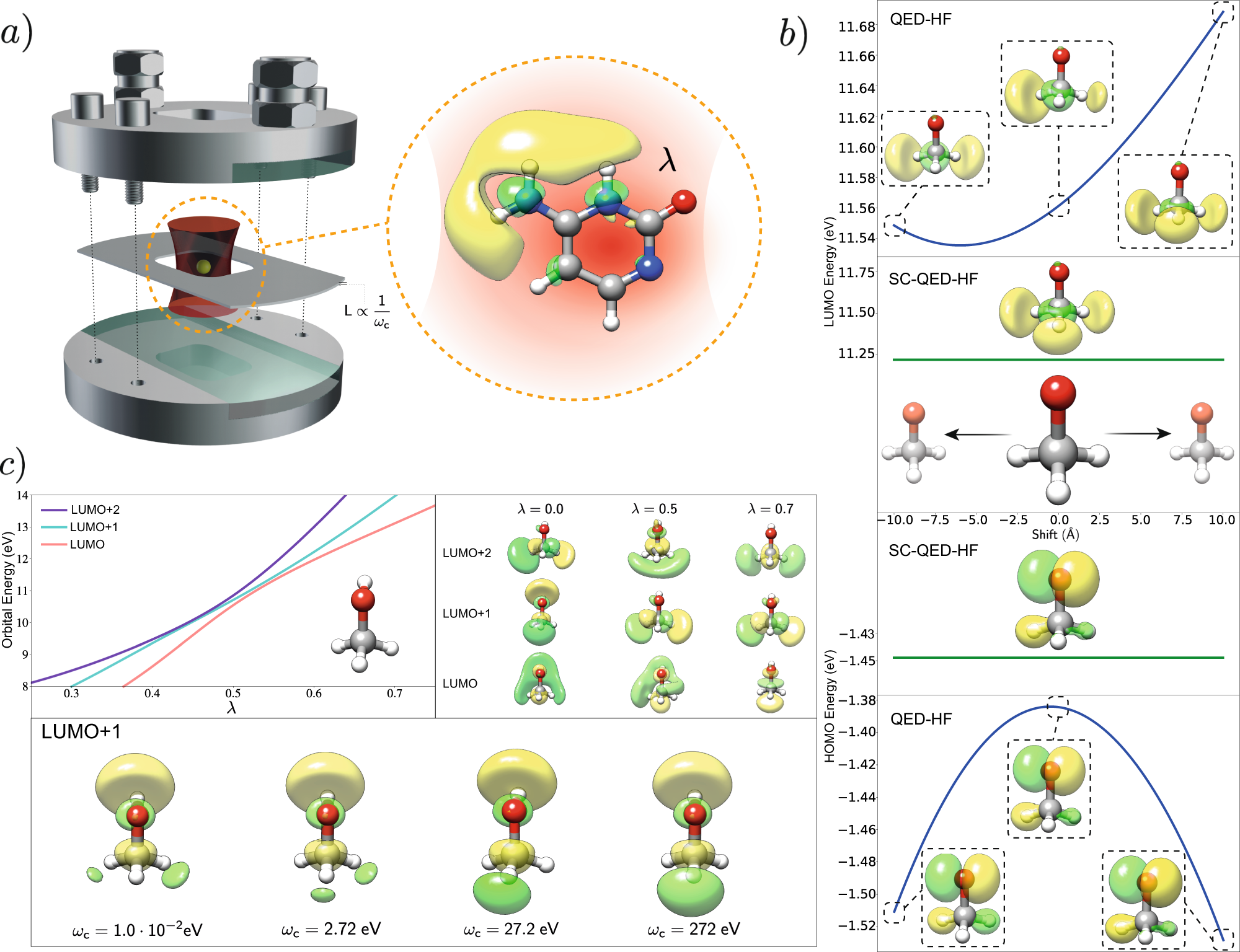
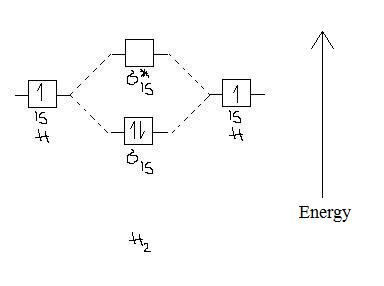
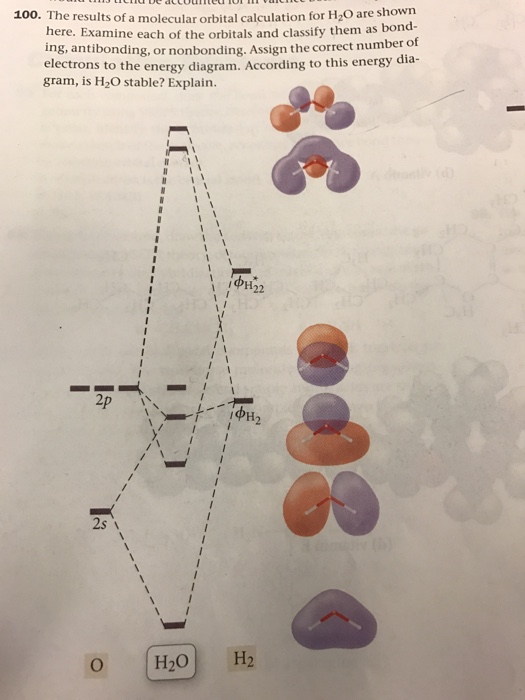
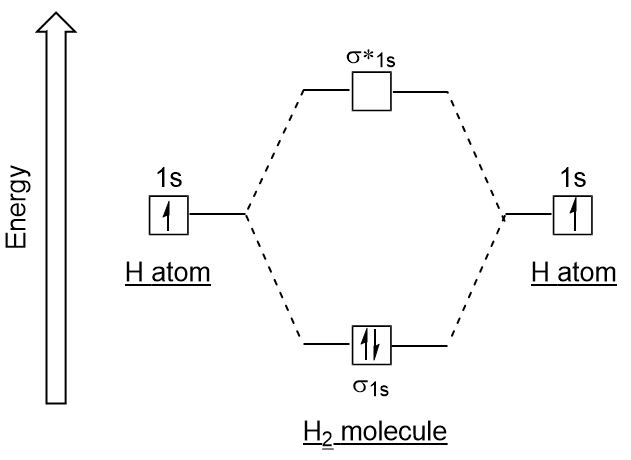
a) Calculate and display the molecular orbitals of NO-. Show how the reaction of NO- and H+ can be described as a HOMO-LUMO interaction. b) Calculate and display the molecular orbitals of

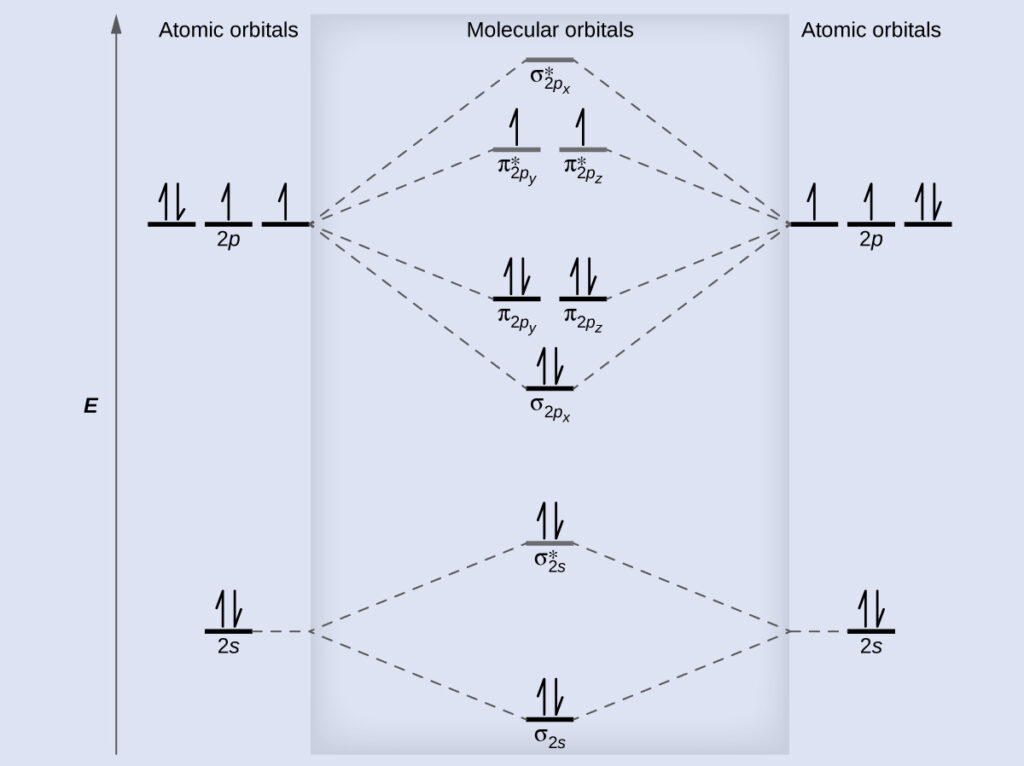
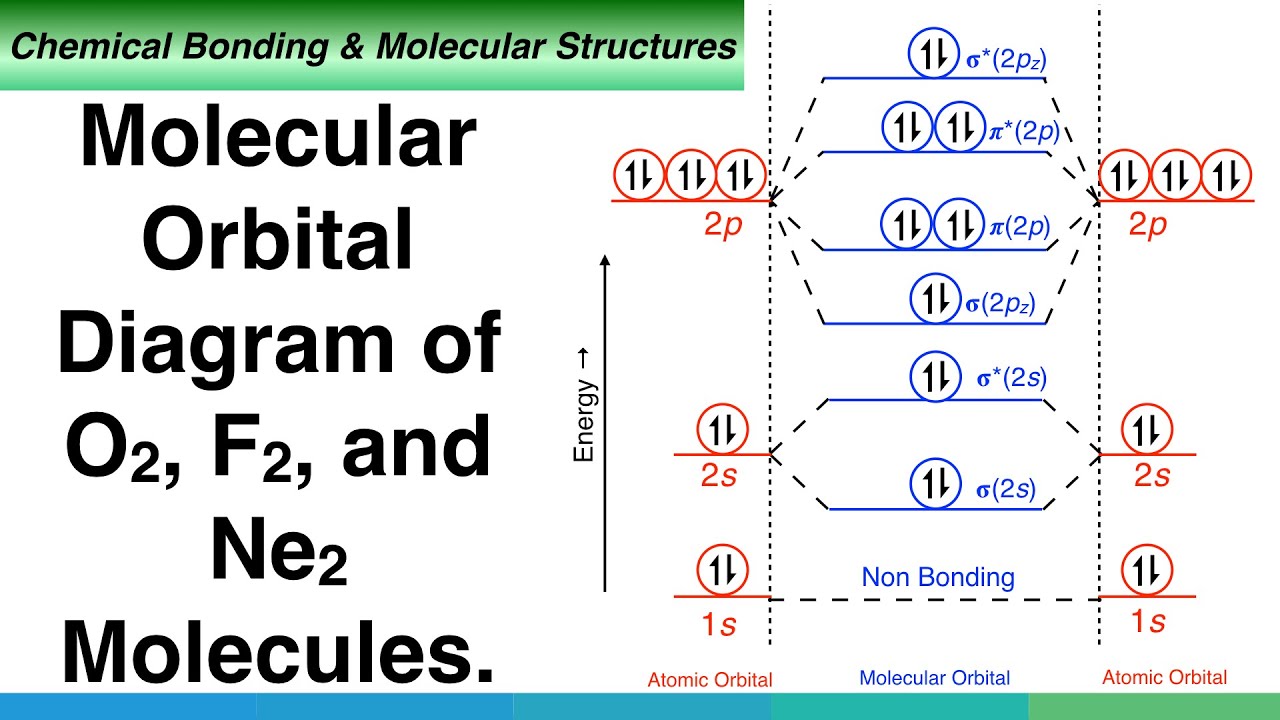
Draw the molecular orbital diagram of O2 and calculate the bond order. Is O2 diamagnetic or paramagnetic? Explain your answer. | Homework.Study.com
GitHub - ZhangJunQCC/Huckel-Molecular-Orbital-Calculation: A program to perform Huckel molecular orbital caluclations on molecules.

FMODB: The World's First Database of Quantum Mechanical Calculations for Biomacromolecules Based on the Fragment Molecular Orbital Method | Journal of Chemical Information and Modeling

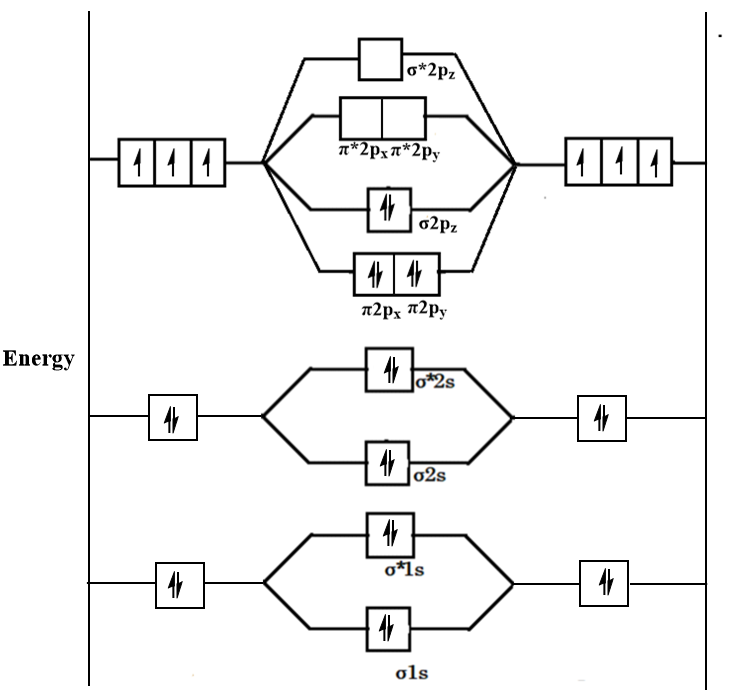
Write the molecular orbital configuration of O(2)^(+) Calculate its bond order and predict its magnetic behaviour.