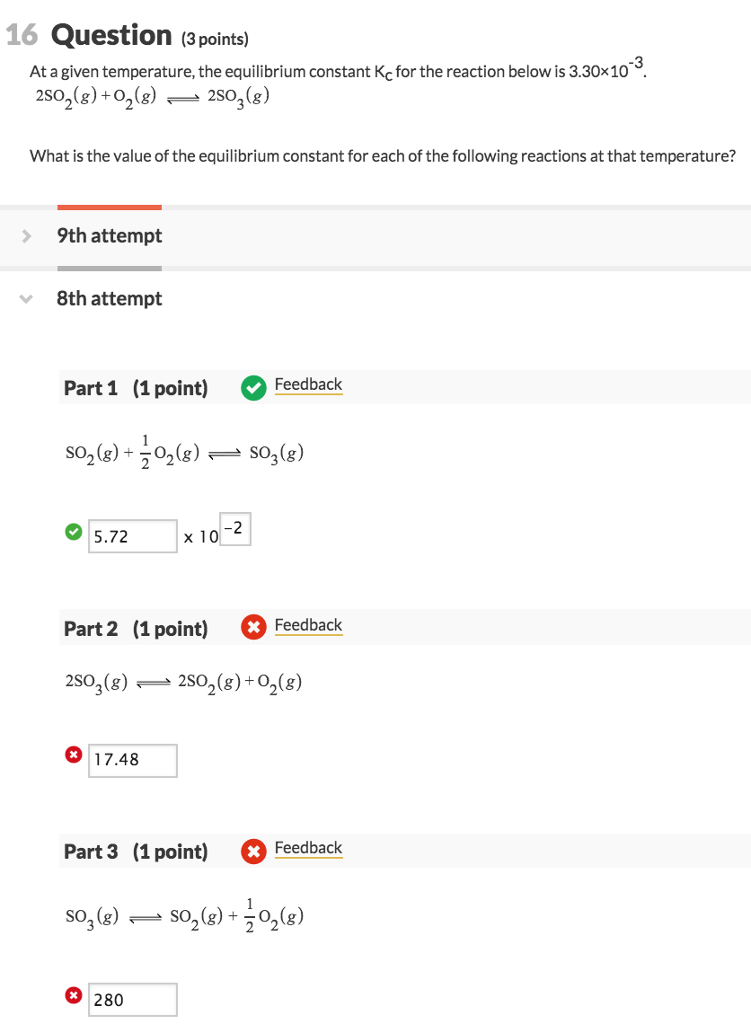
SOLVED: Calculate the Reaction Quotient (Q) for AgCI when 500 mL of 4 x 10-5M AgNO3(aq) is mixed with 500 mL of 2 X 10-SM NaCllaq) and determine whether AgCI will precipitate

Two formulal to calculate number of milli equivalents (mlQ) Numbr of miliequivalents =("weight")/("GEW")xx1000 Numbr of milliequivalents= volume in ml xx Normality of solution 0.25 grams of pure CaCO(3) neutralised 25 ml dilue

q calculate the volume occupied at 270 c and at 2 atm pressure of the gas evolved by 2ml of solid co2 given density of solid co2 is 153 g ml 1

A solution contains 5.6 mL of alcohol mixed with 75 mL of water. Calculate the concentration - YouTube
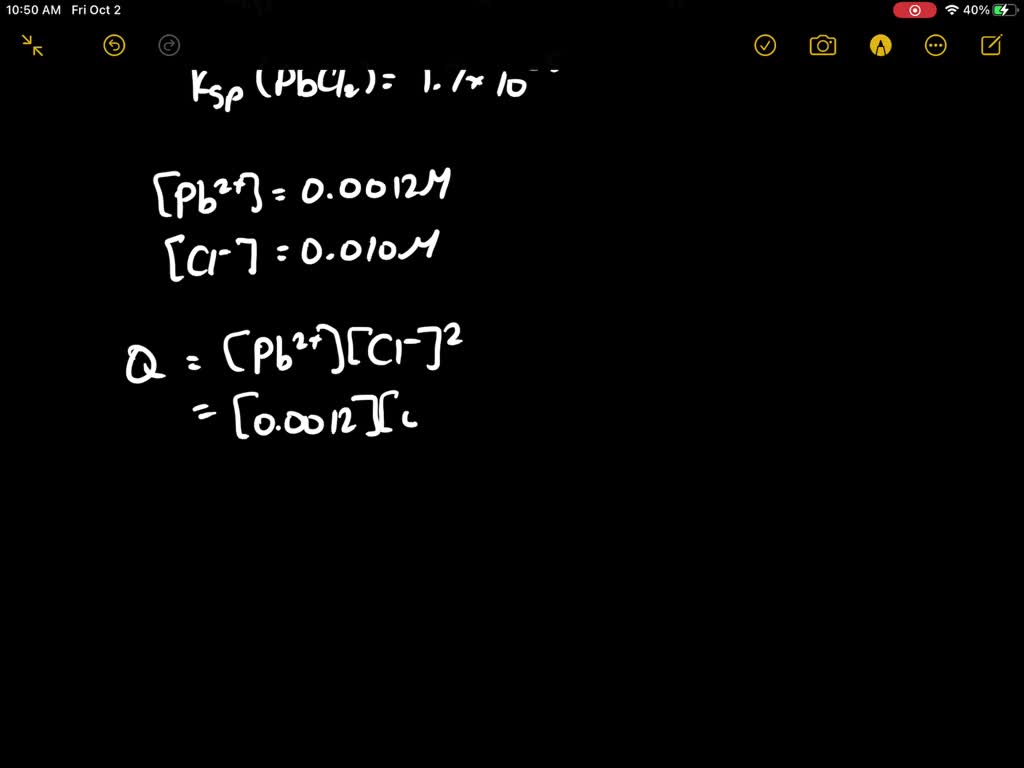
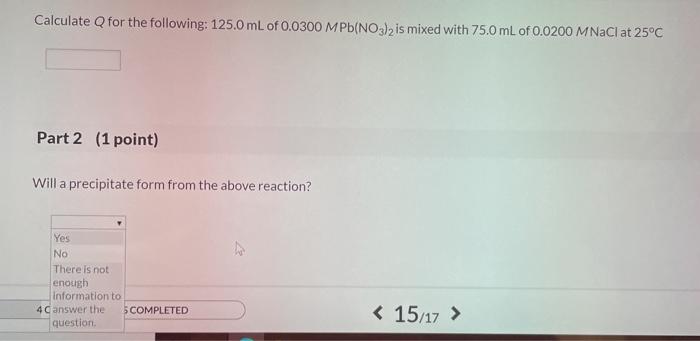
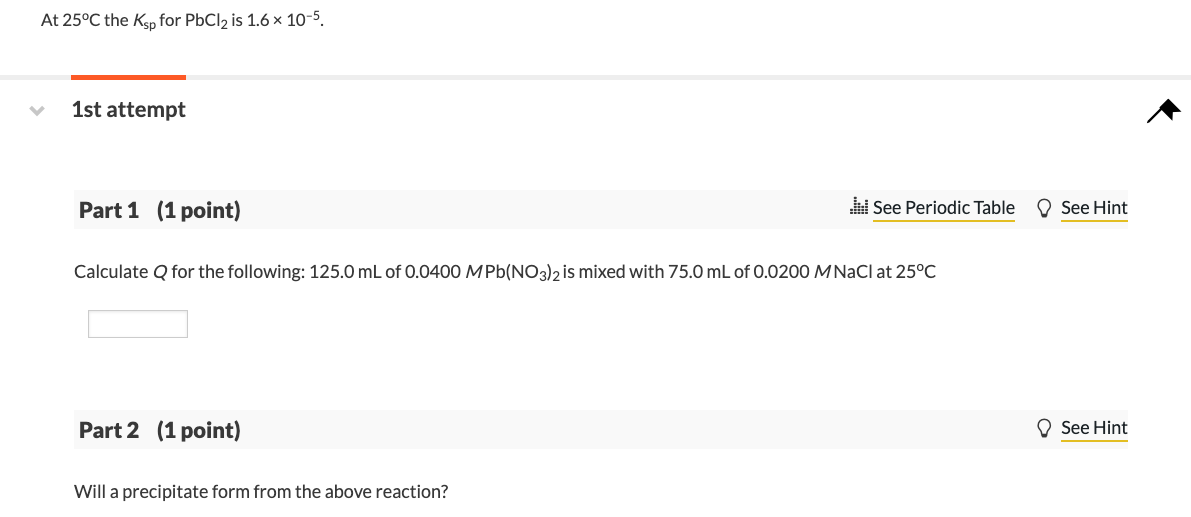
SOLVED: At 25oC the Ksp for PbCl2 is 1.6 × 10–5. A. Calculate Q for the following: 125.0 mL of 0.0800 M Pb(NO3)2 is mixed with 75.0 mL of 0.0200 M NaCl